From MedTech to Mammoths: Insights from Frontier Path’s Fund Launch
I had the pleasure of attending the Frontier Path fund launch, where the GP, Dr. Harneet Bath, was joined by an impressive lineup of speakers, including Ben Lamm of Colossal Biosciences, Dr. Josh Makower of Stanford, Andrey Doronichev of Bioptic, and Dr. Edward Zuckerberg, Chief Dental Officer at Viome and Venture Partner at Frontier Path.
These invite-only fund launches, primarily attended by allocators, are incredibly powerful opportunities to connect with fellow investors, as well as doctors and dentists who have deep expertise in the space. It was also a chance to learn more about Dr. Bath's impressive work in MedTech secondaries, which continue to be hot.
Dr. Bath boasts one of the most remarkable track records, with over 100 investments into high-flyer MedTech companies. His approach drew a large crowd, intrigued by the opportunity to bet on compelling medical technologies that are typically six years into their lifecycle. These companies are often on the verge of being acquired by biotechs that have already expressed interest, offering spectacular returns with a much lower risk profile than traditional venture investing.
At these types of fund launches or annual general body meetings, I often find that you learn just as much from fellow attendees as from the speakers on stage. For me, this was certainly the case at this event, where I had the opportunity to compare notes with others making investments into emerging managers, direct investments, and navigating the complexities of the secondary market.
The secondary space continues to present remarkable discounts on established companies, many of which may still be trapped in the inflated valuations of 2021. Despite only 15% of unicorns avoiding a down round that year, these companies are now trading as secondaries, often at prices far below their peak. The discussions with other investors provided valuable insights into how to make sense of this dynamic market and uncover opportunities in what has become an increasingly attractive segment of the investment landscape.
The first speaker is likely the most talked-about figure in biotech over the last decade: the CEO of Colossal Biosciences, who is leading groundbreaking efforts in de-extinction. Having raised $225M to fund these ambitious projects, he’s working on bringing back species like the mammoth, Tasmanian tiger, and dodo. Yes, you read that correctly.
Key takeaways from Ben Lamm's talk on de-extinction:
1. Innovative Technologies for De-Extinction and Conservation: Colossal is leveraging advanced technologies like gene editing, synthetic biology, and AI to revive extinct species such as the woolly mammoth, Tasmanian tiger, and dodo bird. This effort is not only about bringing back extinct species but also aims to accelerate conservation efforts for endangered species by utilizing genetic tools to restore biodiversity and ecosystems.
2. Sustainable Monetization and Impactful Strategies: Colossal is exploring innovative monetization strategies beyond traditional zoos, focusing on reintroducing species to their natural habitats. Through partnerships with governments, indigenous groups, and private landowners, they are tapping into ecotourism, carbon credits, and biodiversity credits to create sustainable financial models that align with conservation goals.
3. Environmental and Climate Change Benefits: The revival of species like the woolly mammoth could have significant environmental benefits, such as aiding in the preservation of permafrost and mitigating climate change by restoring ecosystems that store carbon. This "Arctic rewilding" concept could help reduce the warming effects of melting glaciers, showcasing the potential secondary benefits of de-extinction efforts in addressing climate change.
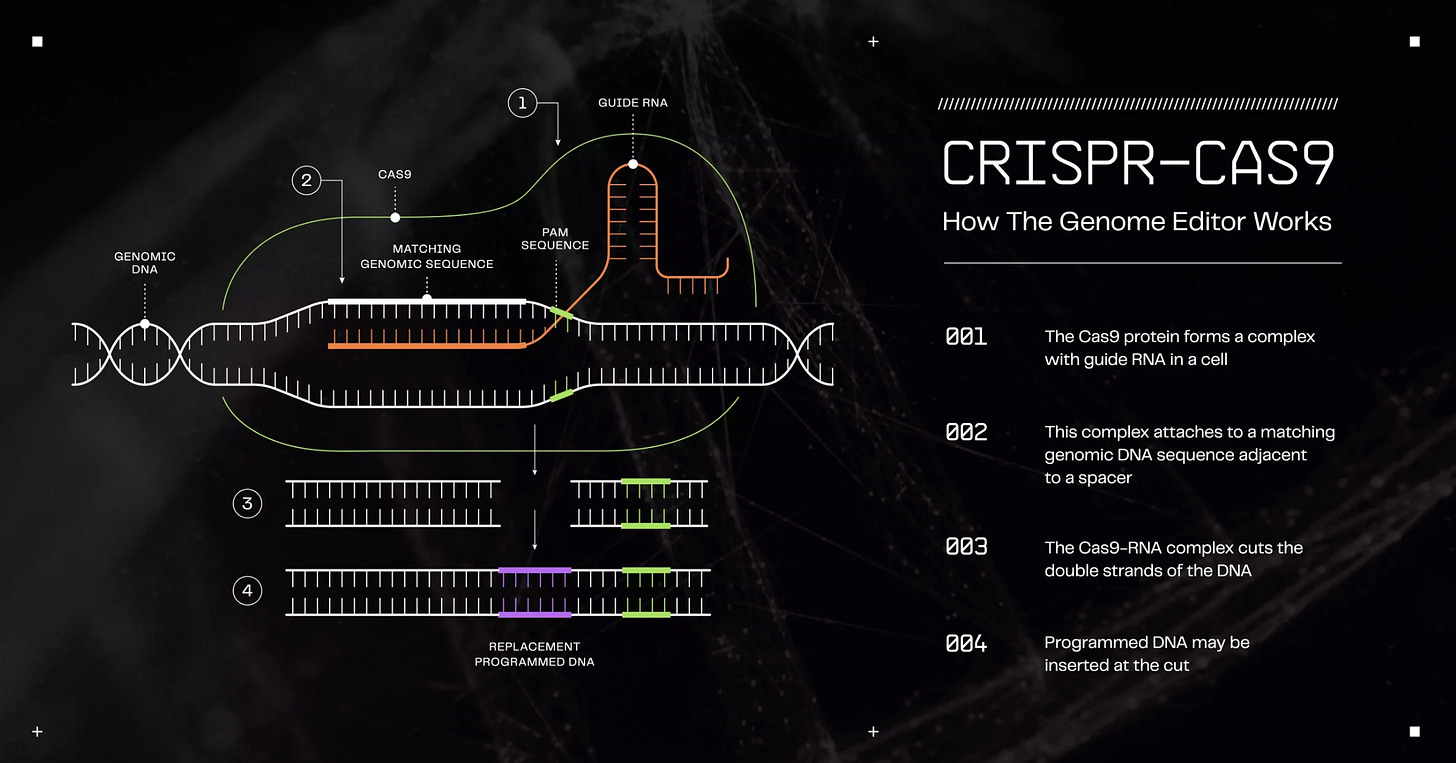
In a conversation with Dr. Bath, Ben Lamm, CEO of Colossal, discussed his entrepreneurial journey, emphasizing his experience in building, scaling, and exiting tech companies to giants like Zynga before diving into de-extinction. Although he lacked a scientific background, Lamm’s fascination with synthetic biology and AI led him to Dr. George Church, inspiring his move to Colossal and its mission in de-extinction. Dr. George Church is a renowned geneticist, Harvard professor, and molecular engineer, known for his pioneering work in genomics, synthetic biology, and de-extinction, and for his contributions to the Human Genome Project and CRISPR technology.
Colossal is focused on the ambitious goal of de-extincting species, starting with the woolly mammoth, the Tasmanian tiger (thylacine), and the dodo bird. These efforts are positioned as part of a broader conservation strategy, as Lamm explained that the company's technology not only seeks to bring extinct species back but also aims to aid in the preservation of endangered ones. The goal is to accelerate conservation efforts through the use of genetic technologies and assist in the restoration of biodiversity. Lamm emphasized the importance of an integrated toolkit that could aid conservation efforts for other endangered species, such as the northern white rhino, which is functionally extinct.
The process of de-extinction, as described by Lamm, involves using advanced genetic techniques to modify and enhance the genomes of related species. For instance, the company is working on editing elephant DNA to bring back the woolly mammoth by utilizing samples from frozen zoos and museum collections. These samples will help create a diverse genetic pool and enable the engineering of traits that resemble those of the extinct species. Additionally, the company is working on creating induced pluripotent stem cells (iPSCs), which can be used to generate egg cells for species like the northern white rhino, circumventing the need to extract eggs from the last remaining females.
Lamm mentioned that Colossal expects to have the first woolly mammoth calves by late 2028. Although the company is on track with its plans, Lamm noted that the 22-month gestation period for elephants means the first mammoths will take some time. However, he expressed optimism that Colossal would likely succeed in bringing back an extinct species before 2028, though it would not be the mammoth.
Throughout the conversation, Lamm discussed the scientific and ethical considerations of de-extinction. He acknowledged the challenges of working with ancient DNA and the limitations in recovering viable genetic material from species that have been extinct for tens of thousands of years. He also reflected on the practical conservation benefits of de-extinction efforts, noting that bringing back keystone species like the woolly mammoth could have positive ecological effects, such as restoring certain ecosystems or helping mitigate climate change. However, Lamm was clear that the focus is not on resurrecting dinosaurs—an idea that often comes up in public discussions of de-extinction—because there is no viable DNA to work with, and the ecological benefits of such an endeavor would be minimal.
Ultimately, Lamm sees Colossal's work as a bridge between synthetic biology, artificial intelligence, and conservation, offering a new avenue to tackle biodiversity loss and environmental degradation. While the ultimate goal is to revive extinct species, Colossal is also building technologies that can contribute to ongoing conservation efforts. This includes creating genetic rescue tools that can help save species that are on the brink of extinction, using technologies that could help restore their populations without the need for invasive interventions.
The public’s fascination with de-extinction and the potential to see long-extinct creatures return fuels Lamm's vision for Colossal, aiming to not only revive extinct species but also create a sustainable future for biodiversity by revolutionizing conservation and restoring ecosystems with cutting-edge science.
Despite the company’s large valuation of over a billion dollars, the complexities of monetizing groundbreaking technology in fields like animal conservation have led them to rethink traditional business models. Initially, Colossal received significant interest from zoos and private collectors, with over 700 orders from zoos worldwide. While this presented a lucrative business opportunity, Lamm highlights that the company's core values didn’t align with just putting animals back in zoos. This led to a pivot toward exploring more sustainable and impactful monetization strategies.
Colossal’s work involves sophisticated biotechnologies, including assisted reproductive technology and gene-editing methods, which have opened up new opportunities far beyond simple exhibition. The company has achieved significant advancements, such as multiplex editing in marsupial cells with over 90% efficiency, which is a breakthrough in the biotechnology field. These innovations extend to areas like plastic degradation, bioluminescence technologies, and early embryonic development, providing Colossal with multiple revenue streams from its intellectual property.
Another monetization strategy that Colossal pursued is reintroducing animals into their natural habitats rather than keeping them in captivity. Lamm explains that working with local governments, indigenous groups, and private landowners to reintroduce species offers a long-term financial benefit through ecotourism, carbon credits, and biodiversity credits. This approach not only aligns with Colossal's values of animal conservation but also provides a higher financial return than the zoo model, while allowing animals to thrive in their natural environment.
In addition, Colossal has explored partnerships with global brands and governments, who have invested substantial sums in saving endangered species. Lamm mentions instances where they have helped shorten the timeline for saving a species from decades to just a few years, offering both ecological and financial benefits. This model appeals to entities with national or global stakes in conservation, as the reduced timeline for species recovery can significantly impact their financial plans and reputation.
The company employs advanced technologies, including computational analysis, synthetic biology, and directed evolution, and collaborates with a network of over 170 scientists and 17 research partners across multiple labs. The first woolly mammoth, estimated to cost mid-eight figures, is expected to be the most expensive species the company works on. Ben contrasts this cost with previous efforts in the field, noting that early de-extinction projects operated on much smaller budgets.
Dr. Bath posed a question regarding the potential to use similar technology to enhance plant life for carbon removal, touching on a broader interest in addressing climate change through bioengineering. Lamm acknowledges this possibility, sharing an example of a related discovery in plastic degradation. The company, Colossal, has spun out a project aimed at breaking down plastic using bacteria, an innovation that, though still in early stages, promises a solution to plastic waste. While Colossal’s current focus is on species revival, Ben envisions the possibility of venturing into plant-related projects in the future.
Lamm then discusses the potential environmental benefits of resurrecting extinct species like the woolly mammoth. One possible secondary benefit is the reduction in the degradation of glaciers due to the presence of large herbivores, such as mammoths, walking across the landscape. This could have an impact on permafrost and carbon storage, especially in areas like Alaska, where permafrost holds more than three times the carbon present in the atmosphere. He explains that large, cold-adapted megafauna, including species like musk oxen and horses, have already demonstrated the ability to lower ground temperatures during the winter months, which could prevent carbon release when the permafrost thaws. Ben highlights the potential of "Arctic rewilding," which may offer a solution to the climate crisis by leveraging ecological restoration efforts involving extinct or near-extinct species.
Finally, the conversation turns to the unexpected challenges Lamm faces in leading a company focused on de-extinction. He shares that one of the biggest hurdles is reprogramming academic scientists to think like product developers. Coming from a software and hardware background, Ben notes that product development in the tech industry is often highly iterative and product-driven, a mindset that can be difficult to instill in researchers more accustomed to theoretical and academic approaches.
Here are the top three key takeaways from Dr. Makower's talk:
1. Biodesign Methodology and Innovation in Healthcare: Dr. Makower emphasizes the importance of a structured process in healthcare innovation, starting with a deep understanding of patient needs and the broader healthcare system. The Biodesign approach trains individuals to create successful, real-world healthcare solutions by aligning technological innovations with practical needs.
2. Impact of AI in Healthcare: Dr. Bath highlights the transformative role of AI in revolutionizing healthcare, particularly in diagnostics, treatment, and drug discovery. While optimistic about AI’s potential, he also addresses significant challenges, including data privacy, accuracy, and liability, urging careful management to avoid unintended consequences.
3. Education, Policy, and Global Health Impact: Bath discusses the importance of training future healthcare innovators and policy influencers. His program not only creates successful startups but also places graduates in impactful policy positions, influencing healthcare systems and regulations to ensure that innovation reaches patients and drives global change, such as addressing maternal health in East Africa.
Dr. Makower, physician, engineer, entrepreneur, and educator, introduces a transformative approach to healthcare innovation through Biodesign, a program that aims to improve patient outcomes by creating innovators and translating ideas from academia to real-world applications. He starts by emphasizing the importance of innovation in healthcare and how many existing healthcare problems remain unsolved, highlighting the need for new solutions. According to Dr. Makower, the key to fostering such innovation lies in training individuals through a structured process, which has been effective in his own career as well as in the creation of multiple successful healthcare startups.
The Biodesign methodology focuses on a process that begins with identifying and listening to the patient, not just to understand their immediate problems, but to also grasp the broader context, including the healthcare system, regulatory environment, and payment structures. By understanding these aspects, innovators can create solutions that are more likely to succeed. This methodology has been instrumental in minimizing failures and ensuring that innovations meet the real needs of the healthcare system. Bath emphasizes that innovation is a skill that can be taught, and through Biodesign, he has successfully trained individuals who had no prior experience in invention.
Dr. Makower’s background, including founding a medical device incubator and starting several successful healthcare companies, reflects the impact of the Biodesign methodology. He outlines that the companies created through this approach span various areas of healthcare, with a few examples of successful innovators. Notable among them is Todd Britton, the inventor of Shockwave, a company revolutionizing angioplasty, and Joel Goldsmith, who contributed to the development of Abbott's glucose monitoring technology. Bath notes that these innovators, all of whom were first-time inventors, have created technologies that continue to transform the healthcare industry, such as a device to stimulate the brain and treat essential tremors, which has gained significant traction.
One of the most impressive aspects of Biodesign is the ongoing success of its graduates. The companies they founded during their training, as well as those they created afterward, have made a significant impact. For instance, one such company, Viz.AI, is revolutionizing the field of radiology with AI technology that detects strokes in brain scans. By bypassing the waiting time for scans to be reviewed, this technology alerts radiologists immediately, enabling faster treatment and saving lives. Such innovations are part of the broader ecosystem that Biodesign has fostered, with these companies collectively touching over 13 million lives.
Biodesign's reach extends beyond its core program to include various educational initiatives. Dr. Makower highlights that the program trains approximately 500 individuals annually, including undergraduates, graduate students, fellows, and faculty members at Stanford. These individuals come from diverse disciplines, including basic scientists who may be working on promising molecules but need guidance on how to apply their work to real-world problems. Through Biodesign, these scientists are trained to think about the practical applications of their research and are encouraged to find innovative solutions that can have an immediate impact on patients' lives.
Additionally, Dr. Makower discusses the broader influence of Biodesign's training, which extends to executives at multinational corporations and faculty at universities around the world. The program not only helps individuals refine their innovation skills but also transforms the way they view their careers. For scientists, the challenge often lies in applying their research to solve specific problems. Biodesign teaches them to focus on the problem first, identify the technology that could address it, and then apply their expertise to create a solution.
Finally, Dr. Makower touches on the future of healthcare innovation and the role of artificial intelligence. He notes that AI plays a significant role in modern healthcare innovation, particularly in areas such as diagnostics and treatment. Dr. Makower’s latest startup, for example, is an AI-based technology, illustrating the growing intersection between healthcare and AI. Through initiatives like Biodesign, Bath believes that the healthcare sector can continue to evolve and improve, driven by a new generation of innovators equipped with the right tools and methodologies to tackle the complex challenges that lie ahead.
Dr. Makower highlighted the global success of a methodology that trains individuals, particularly students, to become innovators in their respective fields. By providing students with structured tools and methodologies, they have been able to create companies and ecosystems that lead to local economic prosperity. He mentions a particularly exciting initiative in East Africa aimed at addressing maternal deaths caused by hemorrhage. A simple, scalable solution has been developed that could potentially save 100,000 lives annually. This low-cost, low-resource innovation can not only benefit East Africa but also have application in developed countries, underlining the cross-pollination of solutions between regions with different resource levels.
A significant part of Dr. Makower's work includes influencing policy to ensure that innovative technologies are accessible to patients. He argues that without the right policies in place—such as reimbursement structures and funding mechanisms for healthcare technologies—innovation cannot truly succeed. His organization works with policymakers by conducting research and training individuals to shape policies at the governmental level. Dr. Makower's program trains students at institutions like Stanford and places them in high-impact policy positions in Washington, D.C., including roles at the Department of Health and Human Services (HHS), the White House, and agencies like the FDA and CMS (Centers for Medicare & Medicaid Services). These efforts aim to ensure that the regulatory environment supports the deployment of new healthcare technologies and treatments.
Dr. Makower asserts that AI will revolutionize healthcare by transforming data-driven decision-making. With its rapid development, AI is already surpassing human abilities in areas like medical exams, indicating its potential to become central to decision-making and diagnostics across healthcare innovation.
Despite AI's rapid advancements, Dr. Makower acknowledges several barriers to its widespread adoption in healthcare. A significant challenge is that AI can sometimes "hallucinate" or generate incorrect facts, which could result in dangerous or misguided healthcare recommendations. This stochastic nature of AI—where it anticipates what comes next based on probabilities—means that it is not always grounded in hard, verifiable facts. This becomes a particularly dangerous issue in healthcare, where inaccurate information can directly impact patient outcomes.
Dr. Makower warns that while AI's ability to process large amounts of data shows promise, its application in healthcare requires careful oversight to avoid unvalidated treatments or diagnoses. He remains optimistic, however, believing that with proper safeguards and validation, AI could transform healthcare by processing increasingly complex information.
A major challenge Dr. Makower highlights is the difficulty in tracking the long-term outcomes of AI in healthcare at a population level. While AI has the potential to revolutionize the industry, we are still in the early stages of understanding its broader impacts, with crucial data often inaccessible or locked down, making it hard to determine if we are creating a truly beneficial tool for patients or unintentionally introducing risks.
Liability is another critical issue that Dr. Makower raises. What happens if an AI system goes "rogue" and causes harm? In a future where AI manages significant portions of healthcare, particularly primary care, the consequences could be devastating. The issue is compounded by the fact that once AI takes over aspects of healthcare, especially in routine or administrative roles, there could be fewer human professionals in the system to step in should something go wrong. The transition to AI-driven systems could also disincentivize medical professionals from pursuing careers in primary care, potentially creating a gap in human healthcare providers when they are needed most.
Despite these challenges, Dr. Makower is also optimistic about the potential of AI to transform medicine. AI's ability to synthesize vast amounts of biological data offers exciting possibilities for drug discovery, diagnostics, and disease treatment. Institutions like the Chan Zuckerberg Initiative are already pioneering AI programs that aim to unlock new understandings of human biology. AI could enable us to imagine and create therapeutic solutions that would have been previously unimaginable. However, as with any new technology, rigorous testing and validation are necessary to ensure its safety and effectiveness.
AI's role in healthcare is already having a profound impact, but its full potential has yet to be realized. For example, AI can simulate biological responses to interventions, helping to design smarter drugs and devices. These simulations could significantly shorten the research and development cycle, speeding up the arrival of new treatments and tools that could revolutionize patient care.
Dr. Makower emphasizes that AI will soon become ubiquitous, much like word processing and online sales, and its widespread use will no longer be a distinguishing factor for businesses or healthcare providers. Instead, the focus will shift to the practical and innovative applications of AI.
He addresses concerns that AI will replace human workers, particularly in healthcare, arguing that AI is intended to augment human abilities, not replace them. At Stanford, AI accelerates innovation by quickly prioritizing healthcare problems and streamlining the review of clinical literature, helping researchers identify knowledge gaps and advance discoveries more rapidly.
However, the integration of AI raises legal and ethical questions, particularly regarding patenting and intellectual property, as the role of AI in generating ideas or solutions remains unclear. Dr. Makower advises caution until clearer legal frameworks are established. While AI is not used directly for disease diagnosis at Stanford, it enhances patient care by simplifying medical records and improving patient engagement.
Dr. Makower offers a balanced perspective on AI's potential in healthcare, recognizing its transformative promise while highlighting challenges such as data privacy, liability, and workforce impacts. As AI becomes more integrated, collaboration among medical professionals, technologists, policymakers, and legal experts will be crucial to maximizing benefits and minimizing risks.
The discussion also touches on the delicate balance educational institutions must strike between fostering innovation and maintaining their educational mission. Dr. Makower advocates for a system where financial interests do not overshadow education and healthcare values, emphasizing the need to protect founders and ensure the long-term success of innovations without exploitation. Ultimately, the goal is to create an ecosystem where education, innovation, and philanthropy work together to drive positive change in healthcare.
Top three takeaways from Andrey Doronichev’s AI drug discovery talk:
1. AI's Role in Revolutionizing Drug Discovery: Doronichev highlights how AI is accelerating the drug discovery process, particularly in the chemistry phase, by optimizing molecule design. This could significantly reduce the cost and time associated with drug development, although challenges remain in the clinical trial phase, where AI's role is less impactful.
2. The Challenges of AI in Drug Discovery: While AI holds immense potential, there are significant hurdles, including the need for better benchmarking, proper validation of AI-generated molecules, and the biological complexity of human testing. Additionally, the pharmaceutical industry’s slow adoption of AI in clinical trials poses a barrier to faster and more efficient drug development.
3. The Growing Role of Tech Giants in Pharma: Doronichev predicts that companies like Google, Meta, and Amazon will play a major role in the future of AI-driven drug discovery, potentially overtaking traditional pharmaceutical companies. This shift requires mastering key elements such as talent, data, and compute power, and will likely spur new partnerships between AI experts and biotech firms to de-risk investments and foster innovation.
Andrey Doronichev, the founder of Bioptic, presented a comprehensive overview of how artificial intelligence is rapidly transforming the field of drug discovery. Reflecting on his experience at Google, Doronichev shared how his involvement with the team behind the groundbreaking "attention is all you need" paper, which played a pivotal role in AI's development, influenced his perspective on AI's potential. As AI becomes increasingly integral across industries, Doronichev emphasized its growing impact on drug discovery, an area traditionally known for its complexity and high costs.
Bioptic, Doronichev's company, leverages AI to accelerate drug discovery, a process historically known for being slow and expensive. He described drug discovery as a multi-step endeavor, spanning biology, chemistry, and medicine. The journey starts with understanding biological mechanisms and identifying proteins involved in bodily processes, which is followed by designing or discovering molecules. Here, AI plays a key role by optimizing molecule design through advanced computational methods, making the process more efficient and cost-effective.
Doronichev highlighted the scale of the pharmaceutical industry, noting that healthcare is a $4 trillion sector, with $600 billion dedicated to pharmaceuticals. Drug discovery itself accounts for $65 billion of this, and the use of AI is beginning to claim a portion of that market. Projections suggest AI's role in drug discovery will grow faster than the broader pharma market, which is already struggling with high costs and inefficiencies.
The drug discovery process is not only expensive but time-consuming, often requiring 10 years and upwards of $1 billion to bring a single drug to market. Doronichev explained that the process involves stages like biology research, molecule design, and clinical trials for FDA approval. Clinical trials are the most costly phase, and while AI can streamline molecule design, it does not directly address the expensive and time-consuming nature of clinical trials, which remain a significant challenge.
Acknowledging the criticism that AI's role in drug discovery is limited to accelerating the chemistry phase, Doronichev argued that improving this stage can still save billions of dollars. He likened the process to shortening a car ride to the airport—while the trip may not solve all the challenges, reducing the time and cost of the chemistry stage offers significant value. He contended that AI’s impact on the chemistry phase could create substantial long-term savings for the industry.
Doronichev also discussed the risks involved in drug discovery, particularly the declining probability of success at each stage. Only about 30% of drugs progress from preclinical to clinical trials, and while the chances improve slightly as drugs advance through the stages, the overall likelihood of success remains low. Despite these challenges, the financial stakes are high, with some estimates placing the cost of developing a drug at $1.5 billion to $4 billion, underscoring the need for more efficient approaches.
Comparing the tech industry’s Moore's Law to the pharmaceutical sector, Doronichev observed a stark contrast. While computational power has rapidly improved in tech, the efficiency of pharmaceutical research and development (R&D) has slowed and become more costly over time—a trend he called "The Roe's Law." This backward trend highlights the urgent need for AI to help reverse inefficiencies in drug discovery and R&D processes, which have become increasingly burdened by high costs and delays.
In his conclusion, Doronichev emphasized AI's transformative potential in drug discovery, particularly in the chemistry phase, where it can optimize the design of molecules. However, he acknowledged that challenges remain, especially in the clinical trial phase, where AI's application is less pronounced. Despite these hurdles, he expressed optimism that AI platforms like Bioptic will revolutionize the drug discovery process, ultimately leading to faster and more efficient development of new drugs.
Doronichev further discussed the challenges and advancements in AI-driven drug discovery. He began by addressing issues such as poor benchmarking and the lack of discipline in machine learning (ML) practices within the field. Doronichev highlighted how improper validation often results in AI-generated molecules that fail in intellectual property (IP) hearings. This issue is compounded by the overlap of training and benchmarking data in ML models, which can lead to inaccurate results and slow progress.
Doronichev also pointed out that while AI can speed up initial drug development stages, the synthesis and testing of molecules still take considerable time. He noted the potential for innovation through the development of chips for on-site testing and industrial robots to automate processes. However, the biological complexity of human testing remains a significant barrier, as drugs that perform well in lab conditions or animal models may not have the same success in human clinical trials due to the unpredictable nature of the human body.
Despite these obstacles, Doronichev remained optimistic about AI's potential, particularly in decoding biology as a language. He compared proteins to words in a book and suggested that large language models (LLMs) could help decode this "biological language." AI could ultimately help unlock new insights into proteins and their functions, aiding drug discovery efforts. Doronichev also noted the Nobel Prize in Chemistry awarded to AI scientists for their work on protein structures, a milestone that is already reshaping the drug discovery landscape.
Doronichev further explored the integration of AI with traditional tools like physics-based simulations, noting how AI-driven approaches could create billion-dollar companies by improving drug discovery processes. He expressed confidence that companies like Bioptic would be at the forefront of this transformation, using AI to design more effective drugs at a faster pace. This shift to AI in drug discovery represents a major opportunity for innovation within the pharmaceutical industry.
Doronichev also discussed the intersection of AI and pharmaceutical companies, highlighting how AI-driven startups could evolve to become vertically integrated drug discovery companies. He pointed to companies like Google, which is making significant strides through its spin-off, Isomorphic Labs, as an example of what the future of AI-first pharmaceutical companies might look like. In contrast, large pharmaceutical companies like Pfizer may struggle to keep up with this evolution, as the required expertise in data, talent, and compute power is increasingly found in tech companies rather than traditional pharma.
Doronichev identified the challenge of selecting the right AI platform in drug discovery, with many companies claiming similar capabilities, making it difficult for startups to choose the best fit. He emphasized the importance of partnerships between AI experts and biotech companies to de-risk investments and accelerate the development of AI-driven solutions. These partnerships can help empower biotech firms with trusted AI tools, speeding up the drug discovery process.
In one such example, Doronichev highlighted a $175 million deal in royalties and milestones over 10 targets, where a venture creation fund partnered with an AI company to support drug discovery ventures. This partnership underscores the importance of AI in driving innovation within the pharmaceutical industry, although Doronichev stressed that scientists remain central to the process, likening them to the drivers of a race. The right AI platform acts as the vehicle for success, but the scientists are the ones steering the course.
Reflecting on AI's role in clinical trials, Doronichev acknowledged that while AI is revolutionizing early stages of drug discovery, its impact on clinical trials is still limited. Data for clinical trials is more challenging to obtain, particularly when it comes to human-specific data. Emerging technologies like digital twins, which are virtual representations of a person’s biology, may hold promise for future clinical trials, but these solutions are still in development.
Finally, Doronichev discussed the role of open research and AI platforms offered by companies like Nvidia, which are providing biotech firms with access to AI models for different stages of drug discovery. While open research is valuable, Doronichev predicted that proprietary models will become more important as competition intensifies in the AI drug discovery field. These proprietary systems, which can fine-tune AI models for specific drug discovery targets, will be key differentiators in the industry, offering biotech firms a competitive edge.
Here are the top three takeaways from Dr. Zuckerberg's talk:
1. Early Adoption of Technology in Dentistry: Dr. Zuckerberg's career demonstrates the transformative power of adopting technology early, from using an Atari 800 for online banking in the 1980s to pioneering innovations in dental care, including intraoral cameras, digital X-rays, and custom Electronic Health Records. His commitment to integrating technology into dental practices not only improved efficiency but also enhanced patient care.
2. Link Between Oral Health and Systemic Diseases: Dr. Zuckerberg emphasized the growing evidence that periodontal disease is linked to systemic conditions like heart disease, diabetes, and dementia. This connection has become more scientifically supported over time, leading to greater awareness of the importance of oral health in overall well-being.
3. Innovation in Dental Technology and Thought Leadership: Dr. Zuckerberg's contributions to dental technology startups and his thought leadership through articles and consultations highlight his role in advancing the field. His efforts to integrate technology into dental practices have influenced both the management of dental care and the future of dental practice innovation.
Dr. Zuckerberg has long been recognized as an early adopter of technology, particularly in the field of dentistry, where he embraced Electronic Medical Records (EMR) long before they became mandatory. His career has been marked by a consistent drive to integrate technological advancements into his practice, aiming to improve both patient care and operational efficiency.
Dr. Zuckerberg's interest in technology began in the 1980s, well before it became mainstream. He was using computers to streamline both his professional and personal tasks, and in 1981, he was already managing online banking activities like paying bills electronically using an Atari 800 computer—a cutting-edge approach for its time. Despite his wife’s initial resistance to the new technology, preferring traditional methods, Dr. Zuckerberg saw its transformative potential.
By 1985, he deepened his commitment to integrating technology into his work by investing in an IBM PC XT, a device equipped with a 10 MB hard drive and 512 KB of RAM. This investment proved pivotal, allowing him to explore innovations in dental care technology and solidifying his dedication to staying at the forefront of advancements in his field.
Throughout his career, Dr. Zuckerberg was dedicated to modernizing dental practices. In the early 1990s, he adopted intraoral cameras and lasers, followed by the introduction of digital X-rays in 1998, all of which improved efficiency and patient care. In 2004, frustrated by the lack of suitable Electronic Health Record (EHR) systems, he developed his own custom solution to enhance patient data management and tracking.
Dr. Zuckerberg's commitment to innovation extended beyond EHRs. He also embraced Computer-Aided Design (CAD) and Computer-Aided Manufacturing (CAM) technologies, enabling him to offer same-day crowns to patients. This advancement significantly reduced wait times and enhanced the patient experience.
Dr. Zuckerberg’s influence in the dental community grew when he moved to California to be closer to his grandchildren. This move allowed him to broaden his network and forge connections with key figures in the field. One notable partnership was with Howard Farran, founder of Dentaltown, a leading online dental platform. Farran's vision of building a global dental network aligned with Dr. Zuckerberg’s expertise, and he played a crucial role in expanding the platform’s reach by leveraging Facebook’s tools.
In addition to his direct contributions to the field, Dr. Zuckerberg has shared his expertise through writing and thought leadership. He penned articles for Dentaltown, focusing on the integration of technology into dental practices, and his insights on the necessity of a Facebook presence for dental offices opened doors to consulting opportunities and speaking engagements at dental conferences, further cementing his reputation as a key influencer in the industry.
For over 14 years, Dr. Zuckerberg has been committed to helping dental technology startups thrive, with his work at companies like Perceptive demonstrating his dedication to integrating technology into dentistry and shaping the field's future. A central focus of his efforts is the vital connection between oral health and systemic diseases, particularly how periodontal disease can contribute to conditions like heart disease, diabetes, and dementia. He highlights how the inflammation caused by gum disease allows bacteria to enter the bloodstream, which can then spread to other parts of the body, leading to serious health complications.
Overall, the event was one of the most polished fund launches I've attended, with a top-tier group of attendees for networking and socializing at the impressive Shashi in Mountain View. I’m confident that big things are ahead for Dr. Bath’s fund, especially with Dr. Zuckerberg serving as a venture partner, acting as a powerful catalyst. The core processes that have driven Dr. Bath’s past successes are likely to continue delivering strong results.
Mr. Mowry is the Managing Partner of Palo Alto Partners. He serves as the CIO of a single family office, is an emerging manager, and helps other emerging managers through Coolwater, Kauffman, or in LA by targeting investors who are most aligned with their fund thesis.